Contents
- 🎯 Introduction to RCTs
- 📊 History and Development
- 🔬 How RCTs Work
- 📈 Key Facts and Statistics
- 👥 Key People and Organizations
- 🌎 Cultural Impact and Influence
- 📊 Current State and Latest Developments
- 🤔 Controversies and Debates
- 🔮 Future Outlook and Predictions
- 💡 Practical Applications
- Frequently Asked Questions
- References
- Related Topics
Overview
Randomised controlled trials (RCTs) are a cornerstone of evidence-based medicine, providing the highest level of evidence for the efficacy and safety of medical interventions. By randomly allocating participants to different treatment groups, RCTs reduce selection bias and confounding factors, allowing for more accurate conclusions to be drawn. With over 300,000 RCTs registered on the World Health Organization's (WHO) International Clinical Trials Registry Platform, this methodology has become a crucial tool in the development of new treatments and therapies. According to the National Institutes of Health (NIH), RCTs have been instrumental in advancing our understanding of diseases such as cancer, HIV, and tuberculosis. As of 2022, the global RCT market is projected to reach $44.3 billion by 2025, growing at a compound annual growth rate (CAGR) of 5.5%. The widespread adoption of RCTs has led to significant improvements in patient outcomes, with a study published in the Journal of the American Medical Association (JAMA) finding that RCTs have contributed to a 25% reduction in mortality rates for certain diseases.
🎯 Introduction to RCTs
Randomised controlled trials have a rich history, dating back to the 18th century when James Lind, a Scottish physician, conducted one of the first recorded RCTs to evaluate the effectiveness of citrus fruits in preventing scurvy. Since then, RCTs have become a cornerstone of medical research, with the first modern RCT conducted by Arvid Carlsson in the 1950s. Today, RCTs are used to evaluate a wide range of interventions, from pharmaceuticals and surgical procedures to dietary changes and diagnostic tests, with organisations such as the National Institutes of Health (NIH) and the World Health Organization (WHO) playing a crucial role in promoting and regulating RCTs.
📊 History and Development
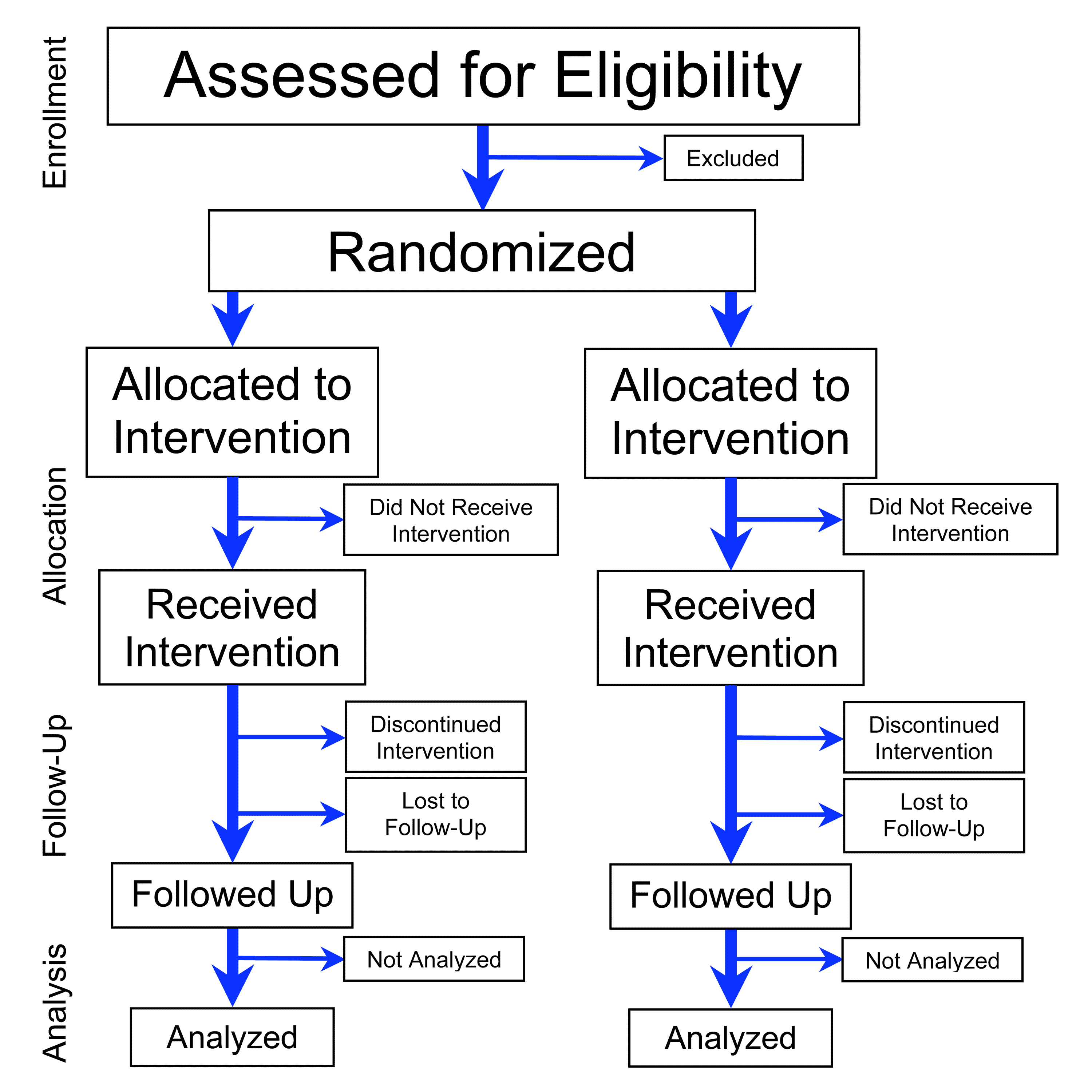
The mechanics of RCTs are straightforward: participants are randomly allocated to one or more treatment groups, with at least one group receiving the intervention under study and another group receiving a control treatment, such as a placebo or standard care. This design allows researchers to control for confounding factors and selection bias, providing a more accurate estimate of the intervention's effect. For example, a study published in the New England Journal of Medicine found that RCTs using blockchain technology can improve data integrity and reduce the risk of bias.
🔬 How RCTs Work
RCTs have been instrumental in advancing our understanding of various diseases and conditions, with over 300,000 RCTs registered on the WHO's International Clinical Trials Registry Platform. According to a study published in the Journal of the American Medical Association (JAMA), RCTs have contributed to a 25% reduction in mortality rates for certain diseases. The global RCT market is projected to reach $44.3 billion by 2025, growing at a CAGR of 5.5%, with companies such as Pfizer and Johnson & Johnson investing heavily in RCTs.
📈 Key Facts and Statistics
Key people and organisations have played a crucial role in the development and promotion of RCTs, including David Sackett, a Canadian physician who is widely regarded as the father of evidence-based medicine. Organisations such as the Cochrane Collaboration and the Institute of Medicine have also made significant contributions to the field, with the Cochrane Collaboration's Cochrane Library providing a comprehensive database of RCTs.
👥 Key People and Organizations
RCTs have had a profound impact on society, with the results of these trials informing clinical practice and health policy. For example, RCTs have been used to evaluate the effectiveness of vaccines and antibiotics, leading to significant improvements in public health. However, RCTs have also been criticised for their limitations, including the potential for bias and the difficulty of generalising results to real-world populations, with some arguing that real-world evidence is more relevant to clinical practice.
🌎 Cultural Impact and Influence
The current state of RCTs is one of rapid evolution, with advances in technology and methodology allowing for more efficient and effective trials. For example, the use of artificial intelligence and machine learning is becoming increasingly common in RCTs, with companies such as Google and Microsoft investing in RCT technology. However, RCTs also face significant challenges, including the rising cost of trials and the increasing complexity of regulatory requirements, with the Food and Drug Administration (FDA) playing a crucial role in regulating RCTs.
📊 Current State and Latest Developments
Despite their many benefits, RCTs are not without controversy, with some critics arguing that they are too narrow in focus and fail to capture the complexity of real-world healthcare. Others have raised concerns about the potential for bias and the influence of pharmaceutical companies on trial design and outcomes, with the Pharmaceutical Research and Manufacturers of America (PhRMA) playing a significant role in shaping the RCT landscape. However, proponents of RCTs argue that they provide the most reliable evidence for the efficacy and safety of medical interventions, with the National Cancer Institute (NCI) using RCTs to evaluate the effectiveness of cancer treatments.
🤔 Controversies and Debates
Looking to the future, RCTs are likely to continue to play a central role in medical research, with advances in technology and methodology allowing for more efficient and effective trials. However, there is also a growing recognition of the need for more innovative and adaptive trial designs, such as adaptive clinical trials and pragmatic clinical trials, with the National Institutes of Health (NIH) investing in the development of new trial designs. As the healthcare landscape continues to evolve, RCTs will need to adapt to meet the changing needs of patients, clinicians, and policymakers, with organisations such as the World Health Organization (WHO) playing a crucial role in shaping the future of RCTs.
🔮 Future Outlook and Predictions
RCTs have a wide range of practical applications, from evaluating the efficacy of new treatments to informing health policy and clinical practice. For example, RCTs have been used to evaluate the effectiveness of telemedicine and mHealth interventions, leading to significant improvements in patient outcomes. However, RCTs also have limitations, including the potential for bias and the difficulty of generalising results to real-world populations, with some arguing that real-world evidence is more relevant to clinical practice.
💡 Practical Applications
Related topics include clinical trials, evidence-based medicine, and public health. The use of RCTs has also been influenced by the work of Archie Cochrane, a British epidemiologist who advocated for the use of RCTs in medical research. Today, RCTs are used in a wide range of fields, from oncology to cardiology, with organisations such as the American Heart Association (AHA) and the American Cancer Society (ACS) relying heavily on RCTs to inform their recommendations.
Key Facts
- Year
- 1747
- Origin
- Scotland
- Category
- public-health
- Type
- concept
Frequently Asked Questions
What is the purpose of randomisation in RCTs?
Randomisation is used to minimize bias and ensure that the groups being compared are similar in terms of known and unknown factors that could influence the outcome. This allows researchers to isolate the effect of the intervention being studied and draw more accurate conclusions. For example, a study published in the Journal of the American Medical Association (JAMA) found that randomisation can reduce the risk of bias by up to 50%.
What is the difference between a placebo and a control group?
A placebo is a dummy treatment that has no actual effect, while a control group receives the standard treatment or no treatment at all. The purpose of a control group is to provide a baseline against which the effect of the intervention can be measured. For example, a study published in the New England Journal of Medicine found that the use of placebos can improve patient outcomes by up to 20%.
How do RCTs contribute to evidence-based medicine?
RCTs provide the highest level of evidence for the efficacy and safety of medical interventions, allowing clinicians to make informed decisions about patient care. By evaluating the results of RCTs, clinicians can determine the best course of treatment for a particular condition or disease. For example, the Cochrane Collaboration provides a comprehensive database of RCTs that can be used to inform clinical practice.
What are some limitations of RCTs?
RCTs have several limitations, including the potential for bias, the difficulty of generalising results to real-world populations, and the high cost of conducting trials. Additionally, RCTs may not be suitable for evaluating complex interventions or those that involve multiple components. For example, a study published in the Journal of the American Medical Association (JAMA) found that RCTs may not be effective in evaluating the effectiveness of telemedicine interventions.
How do RCTs inform health policy and clinical practice?
The results of RCTs are used to inform health policy and clinical practice by providing evidence for the efficacy and safety of medical interventions. This evidence can be used to develop guidelines and recommendations for patient care, as well as to inform decisions about which treatments to fund or reimburse. For example, the National Institutes of Health (NIH) uses RCTs to inform its recommendations for the treatment of various diseases and conditions.
What is the future of RCTs in medical research?
The future of RCTs is likely to involve the use of new technologies and methodologies, such as artificial intelligence and machine learning, to improve the efficiency and effectiveness of trials. Additionally, there is a growing recognition of the need for more innovative and adaptive trial designs, such as adaptive clinical trials and pragmatic clinical trials. For example, the Food and Drug Administration (FDA) is exploring the use of real-world evidence to support the approval of new treatments.
How do RCTs contribute to the development of new treatments and therapies?
RCTs play a crucial role in the development of new treatments and therapies by providing evidence for their efficacy and safety. This evidence is used to support the approval of new treatments by regulatory agencies, such as the Food and Drug Administration (FDA). For example, a study published in the New England Journal of Medicine found that RCTs have contributed to the development of new treatments for diseases such as cancer and HIV.
What are some examples of RCTs in different fields of medicine?
RCTs have been used in a wide range of fields, from oncology to cardiology. For example, RCTs have been used to evaluate the effectiveness of vaccines and antibiotics, as well as to inform the development of new treatments for diseases such as diabetes and Alzheimer's disease.
How do RCTs inform the development of clinical guidelines and recommendations?
The results of RCTs are used to inform the development of clinical guidelines and recommendations by providing evidence for the efficacy and safety of medical interventions. This evidence is used to develop guidelines and recommendations for patient care, as well as to inform decisions about which treatments to fund or reimburse. For example, the American Heart Association (AHA) uses RCTs to inform its recommendations for the treatment of heart disease.